All data were collected only from the inpatients. In addition, the data on biochemical and hematological indices including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin (TBIL), serum creatinine (SCr), haematocrit (Hct), white blood cell (WBC), and red blood cell were also collected.

Patient demographic characteristics including age, gender, dosage per day (DOSE), and postoperative days (POD) were collected in hospital medical records system. Informed verbal consent was obtained from the patients or their caregivers for blood sampling in addition to those required for routine therapeutic drug monitoring. Approval was obtained from the hospital’s ethics committee for the study. Patients and Data Collectionĭata were collected prospectively from Chinese adult liver transplant patients who had been administered tacrolimus at the Tianjin First Hospital, China, from 2008 to 2011. The effects of various demographic, hematological, and biochemical parameters on tacrolimus disposition were also investigated.Ģ.
#Nonmem covariate analysis serial#
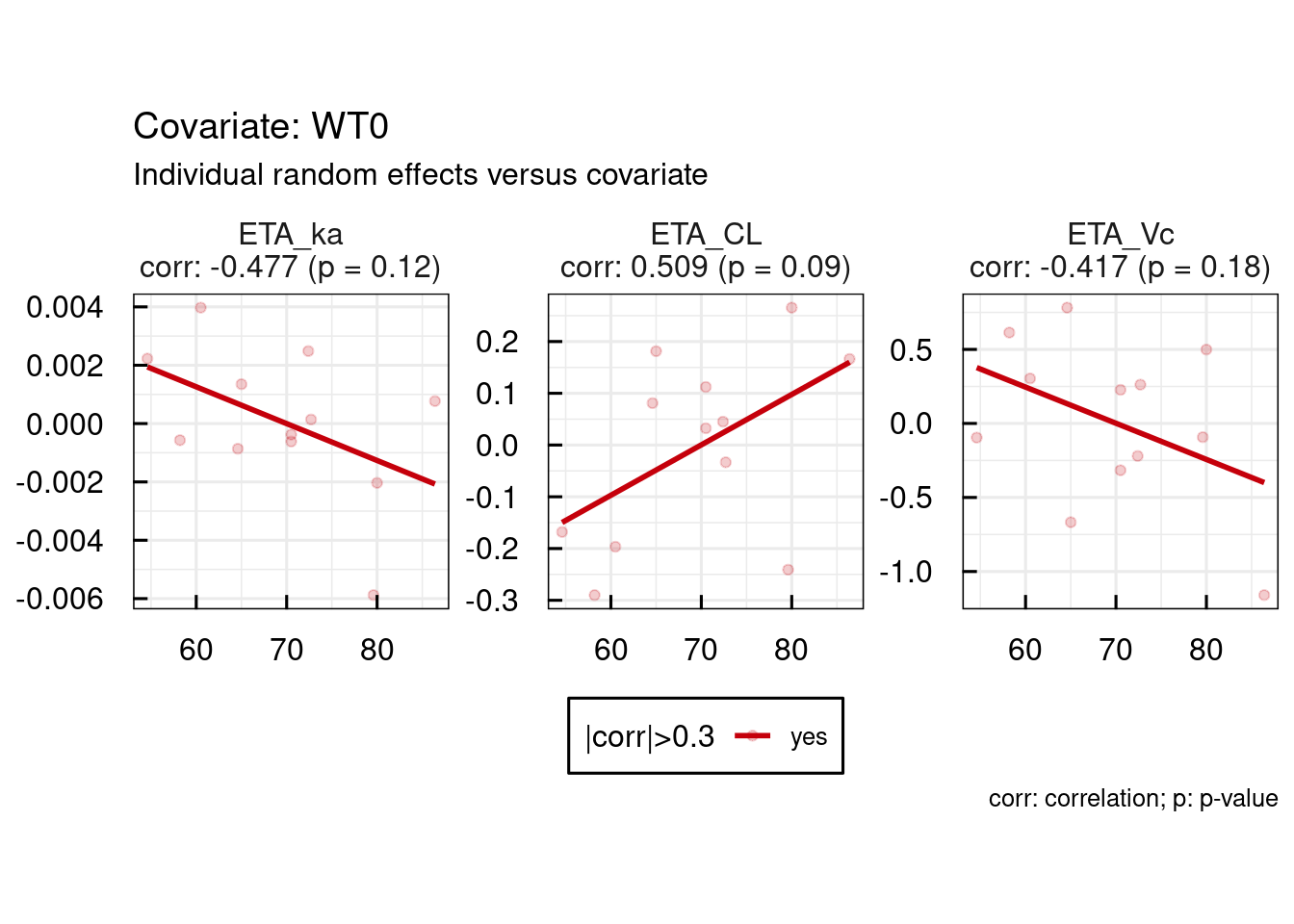
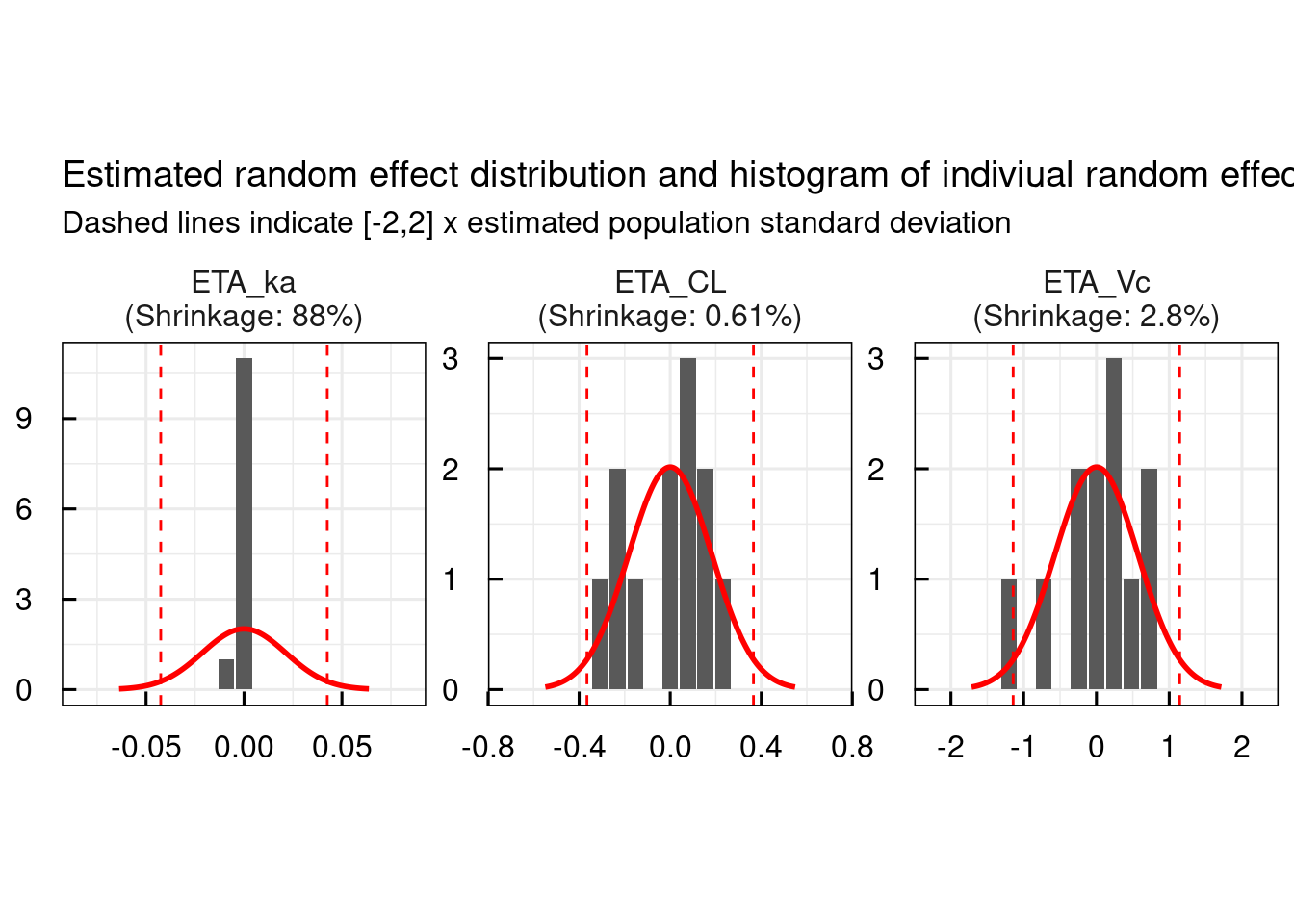
The primary aim of this study was to develop a population pharmacokinetic model to estimate the value of tacrolimus apparent clearance and to assess the intra- and interpatient variability in this parameter in a group of Chinese adult liver transplant recipients using both routine drug monitoring data and serial blood sample during the 12 h interval after administration of oral tacrolimus. There are limited data on the pharmacokinetics of tacrolimus in Chinese adults liver transplant patients. However, the pharmacokinetic parameters observed in different races may not be applicable to Chinese adults liver transplant. From these previous studies, the patients’ hepatic and renal function, body size, age (in pediatrics), time after transplant, and transplant type (whole or cutdown graft) were found to have influence on apparent clearance ( ) of tacrolimus patients’ size and haematocrit level were found to have influence on the apparent distribution volume ( ) of tacrolimus. To date, many population pharmacokinetic studies both in adult and in pediatric liver transplant recipients have been performed. A number of studies have been performed to characterize the pharmacokinetics of tacrolimus in liver transplant recipients. Ī population pharmacokinetic model can be used to predict the dosage regimen most likely to achieve a given target drug concentration based on patient characteristics (covariate values). The area under the concentration-time curve is recognized as a measure for drug exposure. Though trough concentration monitoring is commonly used for dose individualization, the nephrotoxicity may exist when dosing is based on tacrolimus trough concentration alone. Currently, therapeutic drug monitoring is used to adjust the tacrolimus dosage and decrease the adverse effects. Hence, therapeutic drug monitoring and dosage individualization based on patient pharmacokinetics are recommended when tacrolimus is used clinically to reduce the occurrence of adverse events and to optimize treatment outcomes. The administration of tacrolimus is complicated by a narrow therapeutic index, significant inter- and intrapatient variabilities in its pharmacokinetics, and toxicities. Tacrolimus is a macrolide lactone immunosuppressant that is used clinically for the prophylaxis or reversal of organ rejection after organ transplantation. These results could provide the interpretation of the outcome of pharmacokinetics modeling and the impact of covariate tested on individualized tacrolimus therapy. Population pharmacokinetic models of tacrolimus were developed in Chinese adult liver transplant patients. The covariates retained in the final models were postoperative days (POD) and dosage per day (DOSE) on. The interindividual variabilities of these parameters were 16.2%, 163%, 19.7%, 199%, and 74.3%, respectively, and the intraindividual variability of observed concentration was 26.54%. The typical population values of tacrolimus for the pharmacokinetic parameters of apparent clearance ( ), apparent distribution volume of the central compartment ( ), intercompartmental clearance ( ), apparent distribution volume of the peripheral compartment ( ), and absorption rate ( ) were 11.2 L/h, 406 L, 57.3 L/h, 503 L, and 0.723 h −1, respectively. The structural model was a two-compartment model with first-order absorption. Tacrolimus dose and concentration data were obtained from 47 Chinese adult liver transplant recipients, and the data were analyzed using a nonlinear mixed-effect modeling (NONMEM) method.
The aim of this study was to establish population pharmacokinetic models of tacrolimus in Chinese adult liver transplantation patients.
